| Active Ingredient | VORTIOXETINE HYDROBROMIDE |
|---|
| Drug Name | FDA Application No. | Company | Dosage Form;Route | Strength | RLD Strength | Original Approval or Tentative Approval Date |
Exclusivity Expiration (NCE) |
Exclusivity Expiration (ODE) |
Chemical Type |
Review Classification |
Marketing Status |
TE Code |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| BRINTELLIX | (NDA) 204447 | TAKEDA PHARMS USA | TABLET;ORAL | EQ 5MG BASE, EQ 10MG BASE, EQ 15MG BASE, EQ 20MG BASE | EQ 20MG BASE | September 30, 2013 | September 30, 2018 | - | 1 New molecular entity (NME) | S Standard review drug | Prescription | None |
| Parameters | Details |
|---|---|
| Structural Formula |
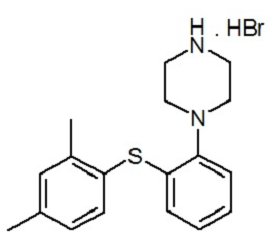 |
| Chemical Name | 1-[2-(2,4-Dimethyl-phenylsulfanyl)-phenyl]-piperazine, hydrobromide |
| CAS No | 508233-74-7 |
| Molecular Formula | C18 H22N2S, HBr |
| Molecular Weight | 379.36 g/mol |
| Appearance | a white to very slightly beige powder |
| Solubility | soluble in methanol and ethanol and slightly soluble in water and aqueous solutions at pH 2.0 to 8.3 |
| Water Solubility | 1.3 mg base/mL |
| Polymorphism | Vortioxetine exhibits polymorphism and appears in four polymorphs. The most thermodynamically stable polymorphic form has been determined and the crystallisation process is designed to consistently deliver this form. |
| pKa (Strongest Acidic) | 9.1 as the free base and 3.0 as the salt |
| pKa (Strongest Basic) | - |
| Log P | 3.48 |
| Identification | HPLC, FTIR and NIR |
| Degradation | - |
| Hygroscopic | non-hygroscopic |
| Photostability study | - |
| Melting Point | >223°C |
| BCS Class | I |
| Manufacture of API | Vortioxetine hydrobromide is manufactured in two well defined synthetic steps, followed by recrystallization and milling. The starting materials used are well defined and commercially available with acceptable specifications. |
| Parameters | Details |
|---|---|
| Indications and Usage | BRINTELLIX is indicated for the treatment of major depressive disorder (MDD). The efficacy of BRINTELLIX was established in six 6 to 8 week studies (including one study in the elderly) and one maintenance study in adults. |
| Dosage and Administration |
The recommended starting dose is 10 mg administered orally once daily without regard to meals. The dose should then be increased to 20 mg/day, as tolerated. Consider 5 mg/day for patients who do not tolerate higher doses TRINTELLIX can be discontinued abruptly. However, it is recommended that doses of 15 mg/day or 20 mg/day be reduced to 10 mg/day for one week prior to full discontinuation if possible The maximum recommended dose is 10 mg/day in known CYP2D6 poor metabolizers |
| Mechanism of action | The mechanism of the antidepressant effect ofvortioxetine is not fully understood, but is thought to be related to its enhancement of serotonergic activityin the CNS through inhibition of the reuptake of serotonin (5-HT). It also has several other activities including 5-HT3 receptor antagonism and 5-HT1A receptor agonism. The contribution of these activities to vortioxetine’s antidepressant effect has not been established. |
| Absorption | The maximal plasma vortioxetine concentration (Cmax) after dosing is reached within 7 to 11 hours postdose (Tmax). Steady state mean Cmax values were 9, 18, and 33 ng/mL following doses of 5, 10, and 20 mg/day. Absolute bioavailability is 75%. |
| Food Effect | No effect of food on the pharmacokinetics was observed. |
| Distribution | The apparent volume of distribution of vortioxetine is approximately 2600 L, indicating extensive extravascular distribution. The plasma protein binding ofvortioxetine in humans is 98%, independent of plasma concentrations. No apparent difference in the plasma protein binding between healthy subjects and subjects with hepatic (mild, moderate) or renal (mild, moderate, severe, ESRD) impairment is observed. |
| Metabolism | Vortioxetine is extensively metabolized primarily through oxidation via cytochrome P450 isozymes CYP2D6, CYP3A4/5, CYP2C19, CYP2C9, CYP2A6, CYP2C8 and CYP2B6 and subsequent glucuronic acid conjugation. CYP2D6 is the primary enzyme catalyzing the metabolism of vortioxetine to its major, pharmacologically inactive, carboxylic acid metabolite, and poor metabolizers of CYP2D6 have approximately twice the vortioxetine plasma concentration of extensive metabolizers. |
| Elimination | Following a single oral dose of [14C]-labeled vortioxetine, approximately 59% and 26% of the administered radioactivity was recovered in the urine and feces, respectively as metabolites. Negligible amounts of unchanged vortioxetine were excreted in the urine up to 48 hours. The presence of hepatic (mild or moderate) or renal impairment (mild, moderate, severe and ESRD) did not affect the apparent clearance of vortioxetine. |
| Peak plasma time (Tmax) | 7 to 11 hours |
| Half life | 66 hours |
| Bioavailability | 75% |
| Age, gender | - |
| DMF | Status | Type | Submit Date | Holder |
|---|---|---|---|---|
| 29849 | A | II | August 15, 2016 | ALP PHARM BEIJING CO LTD |
| 30309 | A | II | March 10, 2016 | MSN PHARMACHEM PRIVATE LTD |
| 31281 | A | II | December 30, 2016 | ALEMBIC PHARMACEUTICALS LTD |
| Parameters | Details | ||||
|---|---|---|---|---|---|
| Strength | 5MG | 10MG | 15 MG | 20 MG | |
| Excipients used | mannitol (110.645MG), microcrystalline cellulose (22.5MG), hydroxypropyl cellulose (4.5MG), sodium starch glycolate (4.5MG), magnesium stearate (1.5MG) | mannitol (104.29MG), microcrystalline cellulose (22.5MG), hydroxypropyl cellulose (4.5MG), sodium starch glycolate (4.5MG), magnesium stearate (1.5MG) | mannitol (97.935MG), microcrystalline cellulose (22.5MG), hydroxypropyl cellulose (4.5MG), sodium starch glycolate (4.5MG), magnesium stearate (1.5MG) | mannitol (91.58MG), microcrystalline cellulose (22.5MG), hydroxypropyl cellulose (4.5MG), sodium starch glycolate (4.5MG), magnesium stearate (1.5MG) | |
| Composition of coating material | Opadry 03B34190 red (4.5MG): hypromellose, titanium dioxide, polyethylene glycol 400, iron oxide red | Opadry 03B22082 yellow (4.5MG): hypromellose, titanium dioxide, polyethylene glycol 400, iron oxide yellow | Opadry 03B33207 orange (4.5MG): hypromellose, titanium dioxide, polyethylene glycol 400, iron oxide red and iron oxide yellow | Opadry 03B250006 red (3.0MG): hypromellose, titanium dioxide, polyethylene glycol 400, iron oxide red | |
| Composition of caspule shell | - | ||||
| Pharmaceutical Development |
During the pharmaceutical development the applicant evaluated different formulations to find the most appropriate combination of excipients and physicochemical and biological properties of the formulation/blend to manufacture the tablet with the best possible tablet characteristics and dissolution rate. The dissolution rate is influenced by the particle size and therefore the active substance is milled to obtain the required particle size distribution throughout the blend. |
||||
| Manufacture of the product | blending, fluid bed granulation, drying, blending, compression and film coating | ||||
| Tablet / Capsule Image |
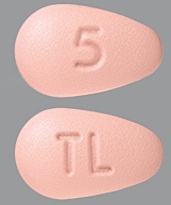
|
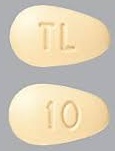
|
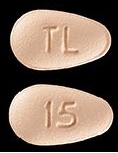
|

|
|
| Appearance | BRINTELLIX is available as immediate-release, film-coated tablets, pink, almond shaped biconvex film coated tablet, debossed with “5” on one side and “TL” on the other side | BRINTELLIX is available as immediate-release, film-coated tablets, yellow, almond shaped biconvex film coated tablet, debossed with “10” on one side and “TL” on the other side | BRINTELLIX is available as immediate-release, film-coated tablets, orange, almond shaped biconvex film coated tablet, debossed with “15” on one side and “TL” on the other side | BRINTELLIX is available as immediate-release, film-coated tablets, red, almond shaped biconvex film coated tablet, debossed with “20” on one side and “TL” on the other side | |
| Imprint code / Engraving / Debossment | “5” on one side and “TL” on the other side | “10” on one side and “TL” on the other side | “15” on one side and “TL” on the other side | “20” on one side and “TL” on the other side | |
| Score | No score | No score | No score | No score | |
| Color | Pink | Yellow | Orange | Red | |
| Shape | TEAR (almond shaped biconvex) | TEAR (almond shaped biconvex) | TEAR (almond shaped biconvex) | TEAR (almond shaped biconvex) | |
| Dimension | 5 × 8mm | 5 × 8mm | 5 × 8mm | 5 × 8mm | |
| Mfg by | - | ||||
| Mfg for | - | ||||
| Marketed by | Takeda Pharmaceuticals (US), H. Lundbeck (US,EU) | ||||
| Distributed by | Takeda Pharmaceuticals (US), H. Lundbeck (US,EU) | ||||
| Application No. | Prod No | Patent No | Patent Expiration | Drug Substance Claim | Drug Product Claim | Patent Use Code | Delist Requested | Link |
|---|---|---|---|---|---|---|---|---|
| N204447 | 1 | 7144884 | January 9, 2023 | Y | Y | U - 1439 | - | Download |
| N204447 | 1 | 8476279 | October 2, 2022 | - | Y | U - 1439 | - | Download |
| N204447 | 1 | 8722684 | June 30, 2031 | Y | Y | - | - | Download |
| N204447 | 1 | 9227946 | June 15, 2027 | - | - | U - 1668 | - | Download |
| N204447 | 1 | 8969355 | June 15, 2027 | - | - | U - 1668 | - | Download |
| USP Apparatus | Speed (RPMs) | Medium | Volume (mL) | Recommended Sampling Times (minutes) | Date Updated |
|---|---|---|---|---|---|
| II (Paddle) | 50 | 0.1 N HCl | 900 | 10, 15, 20, 30 and 45 | May 28, 2015 |
| Label | Link |
|---|---|
| FDA label | Download |
| FDA chemistry review | Download |
| FDA Pharmacology Review(s) | Download |
| FDA Clinical Pharmacology Biopharmaceutics Review(s) | Download |
| FDA BE Recommendation | Download |
| European Public Assessment Report | Download |
| Territory | Brand name / Generic company name | Link |
|---|---|---|
| EU | BRINTELLIX | Download |
| UK | BRINTELLIX | Download |
| US | BRINTELLIX | Download |
| - |
| www.accessdata.fda.gov, www.drugbank.ca, www.ema.europa.eu, www.medicines.org.uk, dailymed.nlm.nih.gov |
