| Active Ingredient | GABAPENTIN ENACARBIL |
|---|
| Drug Name | FDA Application No. | Company | Dosage Form;Route | Strength | RLD Strength | Original Approval or Tentative Approval Date |
Exclusivity Expiration (NCE) |
Exclusivity Expiration (ODE) |
Chemical Type |
Review Classification |
Marketing Status |
TE Code |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| HORIZANT | (NDA) 022399 | XENOPORT INC | TABLET, EXTENDED RELEASE;ORAL | 600MG, 300MG | 600MG (RS), 300MG | April 6, 2011 | - | June 6, 2019 | 1 New molecular entity (NME) | S Standard review drug | Prescription | None |
| Parameters | Details |
|---|---|
| Structural Formula |
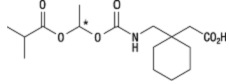 |
| Chemical Name | (1-{[({(1RS)-1-[(2-Methylpropanoyl)oxy]ethoxy}carbonyl)amino]methyl} cyclohexyl) acetic acid |
| CAS No | 478296-72-9 |
| Molecular Formula | C16H27NO6 |
| Molecular Weight | 329.39 |
| Appearance | a white to off-whitecrystalline solid |
| Solubility | a solubility of 0.5 mg/mL in water and 10.2 mg/mL in phosphate buffer (pH 6.3) |
| Water Solubility | 0.5 mg/mL |
| Polymorphism | - |
| pKa (Strongest Acidic) | 5 |
| pKa (Strongest Basic) | - |
| Log P | - |
| Identification | - |
| Degradation | - |
| Hygroscopic | - |
| Photostability study | - |
| Melting Point | Melting onset of about 64°C |
| BCS Class | II |
| Manufacture of API | - |
| Parameters | Details |
|---|---|
| Indications and Usage | HORIZANT (gabapentin enacarbil) Extended-Release Tablets are indicated for the treatment of moderate-to-severe primary Restless Legs Syndrome (RLS) in adults. HORIZANT is not recommended for patientswho are required to sleep during the daytime and remain awake at night. HORIZANT (gabapentin enacarbil) Extended-Release Tablets are indicated for the management of postherpetic neuralgia (PHN) in adults. |
| Dosage and Administration |
Tablets should be swallowed whole and should not be cut, crushed, or chewed. Tablets should be taken with food. HORIZANT is not interchangeable with other gabapentin products because of differing pharmacokinetic profiles. In restless Legs Syndrome, the recommended dosage for HORIZANT is 600 mg once daily at about 5 PM. A daily dose of 1,200 mg provided no additional benefit compared with the 600-mg dose, but caused an increase in adverse reactions. If the dose is not taken at the recommended time, the next dose should be taken the following day as prescribed. In postherpetic Neuralgia, the recommended dosage of HORIZANT is 600 mg twice daily. HORIZANT should be initiated at a dose of 600 mg in the morning for 3 days of therapy, then increased to 600 mg twice daily (1,200 mg/day) on day four. In the 12-week principal efficacy study, additional benefit of using doses greater than 1,200 mg a day was not demonstrated, and these higher doses resulted in an increase in adverse reactions. If the dose is not taken at the recommended time, skip this dose, and the next dose should be taken at the time of the next scheduled dose. |
| Mechanism of action |
Gabapentin enacarbil is a prodrug of gabapentin and, accordingly, itstherapeutic effects in RLS and PHN are attributable to gabapentin. The precise mechanism by which gabapentinis efficacious in RLS and PHN is unknown. The mechanism of action by which gabapentin is efficacious in PHN is unknown but in animal models of analgesia, gabapentin prevents allodynia (pain-related behavior in response to a normally innocuous stimulus) and hyperalgesia (exaggerated response to painful stimuli). Gabapentin prevents pain-related responses in several models of neuropathic pain in rats and mice (e.g., spinal nerve ligation models, spinal cordinjury model, acute herpes zoster infection model). Gabapentin also decreases pain-related responses after peripheral inflammation (carrageenan footpad test, late phase of formalin test), but does not alter immediate pain-related behaviors (rat tail flick test, formalin footpad acute phase). The relevance of these models to human pain is not known. Gabapentin is structurally related to the neurotransmitter gamma-aminobutyric acid (GABA) but has no effect on GABA binding, uptake, or degradation. Gabapentin enacarbil and gabapentin have been tested inradioligand binding assays, and neither exhibited affinity for a number of other common receptor, ion channel, or transporter proteins. In vitrostudies have shown that gabapentinbinds with high affinity to the α2δ subunit of voltage-activated calcium channels; however, the relationship of this binding to the therapeutic effects of gabapentin enacarbil in RLS and PHN is unknown. |
| Absorption |
HORIZANT is an extended-release formulation of gabapentin enacarbil, a prodrug of gabapentin. HORIZANT provides approximately dose-proportional and extended exposure to gabapentin over the range 300 to 6,000 mg. HORIZANT and gabapentin are not interchangeable because the same daily dose of each results in different plasma concentrations of gabapentin. For subjects with PHN taking HORIZANT 600 mg twice daily, the estimated steady state mean Cmax was 5.35 g/mL, mean AUC24 was approximately 109 g*hr/mL, mean Cmin was 3.63 g/mL, and mean peak trough ratio was 1.5. The pathway for absorption of gabapentin enacarbil is believed to include active transport via a proton-linked monocarboxylate transporter, MCT-1. This transporter is expressed at high levels in the intestinal tract and is not saturated by administration of high doses of HORIZANT. Mean bioavailability of gabapentin (based on urinary recovery of gabapentin) for HORIZANT in the fed state is about 75%. Bioavailability under fasting conditions has been estimated by gabapentin urinary recovery to be 42% to 65%. The Tmax of gabapentin after administration of 600 mg of HORIZANT was 5.0 hours in fasted subjects and 7.3 hours in fed subjects. Steady state is reached in 2 days with daily administration. |
| Food Effect | Ina food effect study, the exposure of gabapentin increased by 24%, 34%, and 44% with low, moderate, and high fat meals, respectively. |
| Distribution | Plasma protein binding ofgabapentin has been reported to be <3%. The apparent volume of distribution of gabapentin in subjects receiving HORIZANT is 76 L. |
| Metabolism | After oral administration, gabapentin enacarbil undergoes extensive first-pass hydrolysis by non-specific carboxylesterases primarily inenterocytes and to a lesser extent in the liver, to form gabapentin, carbon dioxide, acetaldehyde, and isobutyric acid. Levels of gabapentin enacarbil in blood are low and transient (2% of corresponding gabapentin plasma levels). Released gabapentin is not appreciably metabolized in humans. Neither gabapentin enacarbil nor gabapentin are substrates, inhibitors, or inducers of the major cytochrome P450 enzymes (CYP1A2, CYP2A6, CYP2B6, CYP2C8, CYP2C9, CYP2C19, CYP2D6, CYP2E1, and CYP3A4). Gabapentin enacarbil is neither a substrate nor an inhibitor of P-glycoprotein in vitro. |
| Elimination | Following hydrolysis of gabapentin enacarbil, the released gabapentin is excreted unchanged by the kidney. Gabapentin renal excretion is believed to involve a component of active secretion via an organic cation transporter (OCT2) present in the kidney. In a human pharmacokinetic study with immediate release 14C gabapentin enacarbil, mean recovery of total radioactivity in urine was 94%, with 5%of the radioactive doserecovered in feces. Apparent oral clearance (CL/F) of gabapentin from plasma after dosing of HORIZANT with food ranged from 6.0 to 9.3 L/hr. Followingoral dosing of HORIZANT, plasma clearance of gabapentin is approximately proportional to creatinine clearance. Renal clearance (CLr) of gabapentin ranged from 5 to 7 L/hr, regardless of food intake or food type. The elimination half-life (t½) of gabapentin ranges from 5.1 to 6.0 hours and is unaltered by dose or following multiple doses of HORIZANT. |
| Peak plasma time (Tmax) | 5.0 hours (fasted) and 7.3 hours (fed) |
| Half life | 5.1 to 6.0 hours |
| Bioavailability | 42% to 65% (fast) and 75% (fed) |
| Age, gender | There are no clinically meaningful differences in pharmacokinetics of HORIZANT between male and female patients. There are no clinically significant differences in pharmacokinetics of HORIZANT between geriatric patients (65 years of age) and younger patients (18 to <65 years of age). However, the pharmacokinetics in geriatric patients may be affected by an age-related decline in renal function. |
| DMF | Status | Type | Submit Date | Holder |
|---|---|---|---|---|
| Not Available | ||||
| Parameters | Details | ||
|---|---|---|---|
| Strength | 300MG | 600MG | |
| Excipients used | colloidal silicon dioxide, dibasic calcium phosphate dihydrate, glyceryl behenate, magnesium stearate, sodium lauryl sulfate, talc and red ferric oxide. | colloidal silicon dioxide, dibasic calcium phosphate dihydrate, glyceryl behenate, magnesium stearate, sodium lauryl sulfate, and talc | |
| Composition of coating material | - | ||
| Composition of caspule shell | - | ||
| Pharmaceutical Development | - | ||
| Manufacture of the product | - | ||
| Tablet / Capsule Image |

|
||
| Appearance | red, with occasional black/grey spots oval-shaped tablets debossed with “GS TF7” | white to off-white, with occasional black/grey spots, oval-shaped tablets debossed with “GS LFG” | |
| Imprint code / Engraving / Debossment | debossed with “GS TF7” and plain on other side | debossed with “GS LFG” and plain on other side | |
| Score | no score | no score | |
| Color | RED | WHITE | |
| Shape | OVAL | OVAL | |
| Dimension | 15mm | 19mm | |
| Mfg by | - | ||
| Mfg for | - | ||
| Marketed by | GlaxoSmithKline (US) | ||
| Distributed by | - | ||
| Application No. | Prod No | Patent No | Patent Expiration | Drug Substance Claim | Drug Product Claim | Patent Use Code | Delist Requested | Link |
|---|---|---|---|---|---|---|---|---|
| N022399 | 1 | 8026279 | November 10, 2026 | Y | Y | - | - | Download |
| N022399 | 1 | 8048917 | November 6, 2022 | Y | Y | U - 1247 | - | Download |
| N022399 | 1 | 8114909 | April 11, 2026 | - | - | U - 1231 | - | Download |
| N022399 | 1 | 8686034 | January 24, 2025 | - | - | U - 1231 | - | Download |
| N022399 | 1 | 8795725 | June 10, 2029 | - | Y | U - 1247 | - | Download |
| N022399 | 2 | 6818787 | November 6, 2022 | DS | DP | - | - | Download |
| USP Apparatus | Speed (RPMs) | Medium | Volume (mL) | Recommended Sampling Times (minutes) | Date Updated |
|---|---|---|---|---|---|
| II (Paddle) | 50 | 10 mM Phosphate buffer at pH 7.4 with 1.0 % SLS | 500 (for 300 mg); 900 (for 600 mg) | 0.5, 1, 2, 4, 6, 8, 12 and 24 hours | January 31, 2013 |
| Label | Link |
|---|---|
| FDA label | Download |
| FDA chemistry review | Download |
| FDA Pharmacology Review(s) | Download |
| FDA Clinical Pharmacology Biopharmaceutics Review(s) | Download |
| FDA BE Recommendation | Download |
| European Public Assessment Report |
| - |
| - |
