| Active Ingredient | ESOMEPRAZOLE MAGNESIUM, TABLET, DELAYED RELEASE |
|---|
| Drug Name | FDA Application No. | Company | Dosage Form;Route | Strength | RLD Strength | Original Approval or Tentative Approval Date |
Exclusivity Expiration (NCE) |
Exclusivity Expiration (ODE) |
Chemical Type |
Review Classification |
Marketing Status |
TE Code |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| NEXIUM 24HR | NDA#207920 | ASTRAZENECA LP | TABLET, DELAYED RELEASE;ORAL | 20MG, EQ 20MG BASE | EQ 20MG BASE | November 23, 2015 | - | - | Type 3 - New Dosage Form | STANDARD | Over-the-counter | None |
| Parameters | Details |
|---|---|
| Structural Formula |
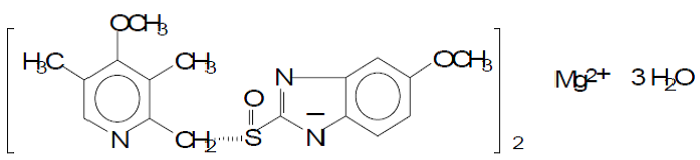 |
| Chemical Name | Bis(5-methoxy-2-{(S)[(4-methoxy-3,5-dimethyl-2-pyridinyl)methyl]sulfinyl}-1H-benzimidazol-1-yl) magnesium |
| CAS No | 119141-88-7 |
| Molecular Formula | (C17H18N3O3S)2Mg x 3 H2O |
| Molecular Weight | 767.2 as a trihydrate and 713.1 on an anhydrous basis |
| Appearance | a white to slightly coloured crystalline powder |
| Solubility | It is soluble in aqueous solutions with pH of 10.0. It contains 3 moles of water of solvation and is slightly soluble in water. The stability of esomeprazole magnesium is a function of pH; it rapidly degrades in acidic media, but it has acceptable stability under alkaline conditions. At pH 6.8 (buffer), the half-life of the magnesium salt is about 19 hours at 25°C and about 8 hours at 37°C. |
| Water Solubility | It is slightly soluble in water. |
| Polymorphism | - |
| pKa (Strongest Acidic) | 9.68 (Predicted) |
| pKa (Strongest Basic) | 4.77 (Predicted) |
| Log P | 0.6 |
| Identification | IR; UV, optical rotation |
| Degradation | Forced degradation studies were conducted by exposing one batch of the active substance to high temperature, acid, base and oxidative conditions. It was only noted a slight increase of impurities, but still within specification limits of the active substance. |
| Hygroscopic | not hygroscopic |
| Photostability study | Photo-stable |
| Melting Point | 155 °C |
| BCS Class | II |
| Manufacture of API | Esomeprazole magnesium trihydrate is synthesized in four main steps using commercially available and well defined starting materials. The final active substance is purified by crystallisation. The chirality of the intermediate has an impact in the final chirality profile of the active substance. The chirality of the active substance is controlled routinely by HPLC on a chiral-AGP column and specific optical rotation. All critical steps have been identified and discussed. The manufacturing process is well described. Adequate in-process controls are applied during the synthesis. |
| Parameters | Details |
|---|---|
| Indications and Usage | Uses treats frequent heartburn (occurs 2 or more days a week) not intended for immediate relief of heartburn; this drug may take 1 to 4 days for full effect |
| Dosage and Administration |
The recommended dose is 20 mg esomeprazole (one tablet) per day. 14-Day Cours e of Treatment - swallow 1 tablet with a glass of water before eating in the morning - take every day for 14 days - do not take more than 1 tablet a day - swallow whole. Do not crush or chew tablets. - do not use for more than 14 days unless directed by your doctor Repeated 14-Day Cours es (if needed) - you may repeat a 14-day course every 4 months - do not take for more than 14 days or more often than every 4 months unles s directed by a doctor - children under 18 years of age: ask a doctor before use. Heartburn in children may sometimes be caused by a serious condition. |
| Mechanism of action |
Esomeprazole is the S-isomer of omeprazole and reduces gastric acid secretion through a specific targeted mechanism of action. It is a specific inhibitor of the acid pump in the parietal cell. Both the R- and S-isomer of omeprazole have similar pharmacodynamic activity. Esomeprazole is a weak base and is concentrated and converted to the active form in the highly acidic environment of the secretory canaliculi of the parietal cell, where it inhibits the enzyme H+K+-ATPase – the acid pump and inhibits both basal and stimulated acid secretion. |
| Absorption | Esomeprazole, the S-isomer of omeprazole, is acid labile and is administered orally as enteric-coated granules. In vivo conversion to the R-isomer is negligible. Peak plasma levels occur approximately 1 to 2 hours after dose. The absolute bioavailability is 64% after a single dose of 40 mg and increases to 89% after repeated once-daily administration. For 20 mg esomeprazole the corresponding values are 50% and 68%, respectively. |
| Food Effect | Food intake both delays and decreases the absorption of esomeprazole although this has no significant influence on the effect of esomeprazole on intragastric acidity. The AUC of esomeprazole magnesium decreased by 33% for a single dose and 26% at steady state on day 5 when administered after food intake compared with the fasting state, based on a single 40 mg dose. Taking into consideration that the PD effect was independent from food intake this is acceptable for the OTC status. |
| Distribution | Total plasma clearance of esomeprazole is about 17 L/h following a single dose and about 9 L/h after repeated administration. The plasma elimination half-life is about 1.3 hours after repeated once-daily dosing. |
| Metabolism | Esomeprazole is completely metabolised by the cytochrome P450 system (CYP). The major part of the metabolism of esomeprazole is dependent on the polymorphic CYP2C19, responsible for the formation of the hydroxy- and desmethyl metabolites of esomeprazole. The remaining part is dependent on CYP3A4, responsible for the formation of esomeprazole sulphone, the main metabolite in plasma. The parameters in this section reflect mainly the pharmacokinetics in individuals with a functional CYP2C19 enzyme, ie, extensive metabolisers. |
| Elimination | Almost 80% of an oral dose of esomeprazole is excreted as metabolites in the urine, the remainder in the faeces. Less than 1% of the parent drug is found in urine. The applicant explains that major metabolites of esomeprazole have no effect on gastric acid secretion. |
| Peak plasma time (Tmax) | 1 to 2 hours |
| Half life | 8 hours |
| Bioavailability | 50% |
| Age, gender | Poor metabolisers are individuals who lack a functional CYP2C19 enzyme and probably mainly catalyses esomeprazole by CYP3A4 (~2.9±1.5% of the western population). After repeated once-daily administration of 40 mg esomeprazole, the mean AUC was approximately 100% higher in poor metabolisers than in subjects having a functional CYP2C19 enzyme (extensive metabolisers). Mean peak plasma concentrations were increased by about 60%. |
| DMF | Status | Type | Submit Date | Holder |
|---|---|---|---|---|
| 17278 | I | II | March 31, 2004 | DR REDDYS LABORATORIES LTD |
| 18559 | A | II | August 3, 2005 | SUN PHARMACEUTICAL INDUSTRIES LTD |
| 18821 | A | II | September 28, 2005 | CIPLA LTD |
| 19192 | I | II | February 21, 2006 | DR REDDYS LABORATORIES LTD |
| 20457 | A | II | April 13, 2007 | LUPIN LTD |
| 21375 | I | II | May 23, 2008 | JUBILANT ORGANOSYS LTD |
| 22335 | A | II | December 7, 2010 | JUBILANT GENERICS LTD |
| 22363 | A | II | December 29, 2008 | CADILA HEALTHCARE LTD |
| 22376 | I | II | December 31, 2008 | DR REDDYS LABORATORIES LTD |
| 23110 | A | II | September 14, 2009 | TORRENT PHARMACEUTICALS LTD |
| 23120 | A | II | September 16, 2009 | MYLAN LABORATORIES LTD |
| 23379 | A | II | December 16, 2009 | HETERO DRUGS LTD |
| 23448 | A | II | May 26, 2010 | JUBILANT GENERICS LTD |
| 24338 | A | II | November 12, 2010 | GLENMARK PHARMACEUTICALS LTD |
| 24901 | A | II | April 20, 2011 | FIS FABBRICA ITALIANA SINTETICI SPA |
| 25049 | A | II | June 9, 2011 | PCAS |
| 26239 | A | II | August 1, 2012 | AUROBINDO PHARMA LTD |
| 27472 | A | II | September 17, 2013 | RAKS PHARMA PVT LTD |
| 27918 | A | II | April 8, 2014 | HEC PHARM CO LTD |
| 27925 | A | II | January 17, 2014 | METROCHEM API PRIVATE LTD |
| 28163 | I | II | March 27, 2014 | PERRIGO API LTD |
| 28462 | A | II | July 8, 2014 | MINAKEM |
| 28523 | A | II | December 2, 2014 | MACLEODS PHARMACEUTICALS LTD |
| 28548 | A | II | July 3, 2014 | SHOUGUANG FUKANG PHARMACEUTICAL CO LTD |
| 28581 | A | II | October 1, 2014 | ALKEM LABORATORIES LTD |
| 29639 | A | II | September 1, 2015 | CIPLA LTD |
| 29992 | A | II | November 4, 2015 | ZHEJIANG JINHUA CONBA BIO PHARM CO LTD |
| 30036 | A | II | April 15, 2016 | UNION QUIMICO FARMACEUTICA SA (UQUIFA SA) |
| 30076 | A | II | December 31, 2015 | MSN LIFE SCIENCES PRIVATE LTD |
| 30175 | A | II | May 31, 2016 | SUN PHARMACEUTICAL INDUSTRIES LTD |
| 30901 | A | II | September 15, 2016 | ZHEJIANG YONGTAI PHARMACEUTICAL CO LTD |
| Parameters | Details | ||
|---|---|---|---|
| Strength | 20MG (US, EU, UK) | 40MG (UK) | |
| Excipients used | corn starch, crospovidone, D&C red no. 27 aluminum lake, FD&C blue no. 2 aluminum lake, FD&C red no. 40 aluminum lake, glyceryl monostearate, hydroxypropyl cellulose, hypromellose, magnesium stearate, methacrylic acid copolymer, mica, microcrystalline cellulose, paraffin, polyethylene glycol, polysorbate 80, sodium stearyl fumarate, sucrose, talc, titanium dioxide, triethyl citrate Sugar spheres (Sucros & Starch) in EU & UK formulation instead of Sucrose. |
corn starch, crospovidone, D&C red no. 27 aluminum lake, FD&C blue no. 2 aluminum lake, FD&C red no. 40 aluminum lake, glyceryl monostearate, hydroxypropyl cellulose, hypromellose, magnesium stearate, methacrylic acid copolymer, mica, microcrystalline cellulose, paraffin, polyethylene glycol, polysorbate 80, sodium stearyl fumarate, sugar spheres (sucrose and maize starch), talc, titanium dioxide, triethyl citrate | |
| Composition of coating material | - | ||
| Composition of caspule shell | - | ||
| Pharmaceutical Development |
The aim of the pharmaceutical development was to obtain gastro-resistant tablets. The active ingredient is a trihydrate of the alkaline magnesium salt of esomeprazole. This active substance is thermodynamically stable, crystalline and non-hygroscopic. The active substance is soluble in aqueous solutions. Tablet weight is 420MG. This medicinal product has been developed to be a gastric acid secretion inhibitor. However, the active substance is rapidly decomposed in acidic media. Therefore, an enteric-coated formulation was developed in order to avoid any decomposition of the active substance. An in vitro dissolution test method including pre-exposure in 0.1 M HCl for two hours followed by an increase of the pH to 6.8 was conducted and evaluated in terms of predictability in vivo. In addition, a bioavailability study was conducted on three tablets with different releases profiles. Moreover, the data form two previous bioavailability studies were included as supportive data. The in vitro dissolution test method provided a level A correlation for formulations having an in vitro dissolution of 70% or less at 30 minutes. |
||
| Manufacture of the product | - | ||
| Tablet / Capsule Image |
.jpg)
|
.jpg)
|
|
| Appearance | A purple colour, oblong, biconvex tablet with "20mG" on one side and "N" on other side. (US) A light pink, oblong, biconvex, coated tablet engraved with ‘20 mg’ on one side and A/EH on the other side. (EU, UK) | A light pink, oblong, biconvex, coated tablet engraved with ‘40 mg’ on one side and A/EI on the other side. | |
| Imprint code / Engraving / Debossment | N;20 MG (US) | A/EI; 40 MG | |
| Score | no score | no score | |
| Color | PURPLE (US), PINK (EU, UK) | light pink | |
| Shape | OBLONG | OBLONG | |
| Dimension | 7 × 14mm | - | |
| Mfg by | Pfizer Consumer Healthcare Ltd (EU), AstraZeneca (US) | AstraZeneca (UK) | |
| Mfg for | AstraZeneca (US) | AstraZeneca (UK) | |
| Marketed by | Pfizer Consumer Healthcare Ltd (EU), AstraZeneca (US) |
AstraZeneca (UK) | |
| Distributed by | Pfizer Consumer Healthcare Ltd (EU), AstraZeneca (US) | AstraZeneca (UK) | |
| Application No. | Prod No | Patent No | Patent Expiration | Drug Substance Claim | Drug Product Claim | Patent Use Code | Delist Requested | Link |
|---|---|---|---|---|---|---|---|---|
| N207920 | 1 | 5900424*PED | November 4, 2016 | - | - | - | - | Download |
| N207920 | 1 | 6369085 | May 25, 2018 | DS | DP | U-1784 | - | Download |
| N207920 | 1 | 6369085*PED | November 25, 2018 | - | - | - | - | |
| N207920 | 1 | 6428810 | November 3, 2019 | - | DP | U-1785 | - | Download |
| N207920 | 1 | 6428810*PED | May 3, 2020 | - | - | - | - | |
| N207920 | 1 | 7411070 | May 18, 2018 | DS | - | - | - | Download |
| N207920 | 1 | 7411070*PED | November 18, 2018 | - | - | - | - |
| USP Apparatus | Speed (RPMs) | Medium | Volume (mL) | Recommended Sampling Times (minutes) | Date Updated |
|---|---|---|---|---|---|
| II (Paddle) | 100 | Acid stage: 0.1 N HCl; Buffer stage: Phosphate Buffer, pH 6.8 | Acid stage: 300; Buffer stage: 1000 | Acid stage: 120; Buffer stage: 10, 20, 30, 45 and 60 | October 20, 2016 |
| Label | Link |
|---|---|
| FDA label | Download |
| FDA chemistry review | Download |
| FDA Pharmacology Review(s) | Download |
| FDA Clinical Pharmacology Biopharmaceutics Review(s) | Download |
| FDA BE Recommendation | Download |
| European Public Assessment Report | Download |
| Territory | Brand name / Generic company name | Link |
|---|---|---|
| EU | Nexium Control | Download |
| UK | Nexium Control Tablet 20 mg & 40 mg | Download |
| US | NEXIUM 24HR | Download |
| Nexium Control Tablet 20 mg & 40 mg are approved in UK wherelse Nexium Control 20MG is approved in EU. Trade dress of 20MG strength available in US & UK/EU is different. |
| www.accessdata.fda.gov, www.drugbank.ca, www.ema.europa.eu, www.medicines.org.uk, dailymed.nlm.nih.gov |
