| Active Ingredient | CABOTEGRAVIR SODIUM |
|---|
| Drug Name | FDA Application No. | Company | Dosage Form;Route | Strength | RLD Strength | Original Approval or Tentative Approval Date |
Exclusivity Expiration (NCE) |
Exclusivity Expiration (ODE) |
Chemical Type |
Review Classification |
Marketing Status |
TE Code |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| VOCABRIA | 212887 | VIIV HLTHCARE | TABLET;ORAL | EQ 30MG BASE | EQ 30MG BASE | January 21, 2021 | January 21, 2021 | _ | Type 1 - New Molecular Entity | PRIORITY | Prescription | None |
| Parameters | Details |
|---|---|
| Structural Formula |
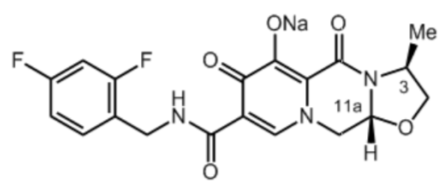 |
| Chemical Name | Sodium (3S,11aR)-N-[(2,4- difluorophenyl)methyl]-6-hydroxy-3-methyl-5,7-dioxo-2,3,5,7,11,11a-hexahydro[1,3]oxazolo[3,2- a]pyrido [1,2-d]pyrazine-8-carboxamide |
| CAS No | 1051375-10-0 |
| Molecular Formula | C19H16F2N3NaO5 |
| Molecular Weight | 427.34 g/mol |
| Appearance | White to almost white crystalline solid |
| Solubility | Slightly soluble in water |
| Water Solubility | - |
| Polymorphism | Cabotegravir exhibits polymorphism and four solid state forms have been identified, of which only two are relevant to the commercial manufacturing process: Form 1 and Form 2. Form 1 was confirmed as the most thermodynamically stable form at ambient conditions and under process relevant conditions. |
| pKa (Strongest Acidic) | 10.4 |
| pKa (Strongest Basic) | -0.7 |
| Log P | 1.4 |
| Identification | IR |
| Degradation | Forced degradation studies have been performed on cabotegravir to identify potential degradation products. Cabotegravir was chemically stable in the solid state under all stressing conditions used in the forced degradation study. There was no significant increase in the total degradation products under any solid-state stress condition. Significant degradation was only observed in solution under acidic, basic and oxidative conditions. However, the degradation pathways observed under solution phase conditions are formed under forcing conditions that are not representative of those that a solid active substance will experience during manufacture or storage. The results from the forced degradation studies demonstrate that the HPLC methods are stability indicating. |
| Hygroscopic | Hygroscopic |
| Photostability study | Photostability was conducted on a pilot batch, according to ICH Q1B. No significant changes was observed. |
| Melting Point | - |
| BCS Class | BCS class II |
| Manufacture of API | The active substance manufacturer has been stated. Cabotegravir manufacturing process consists of 6 steps including a purification step from well-defined starting materials (SMs). These SMs have been sufficiently justified in line with ICH Q11 and are controlled by suitable specifications. There are three isolated intermediates controlled by acceptable specifications. The stereochemistry of cabotegravir is determined either by the starting materials or by the process conditions of the synthetic process. |
| Parameters | Details |
|---|---|
| Indications and Usage | VOCABRIA is a human immunodeficiency virus type-1 (HIV-1) integrase strand transfer inhibitor (INSTI) indicated in combination with EDURANT (rilpivirine) for short-term treatment of HIV-1 infection in adults who are virologically suppressed (HIV-1 RNA less than 50 copies/mL) on a stable antiretroviral regimen with no history of treatment failure and with no known or suspected resistance to either cabotegravir or rilpivirine, for use as: oral lead-in to assess the tolerability of cabotegravir prior to administration of CABENUVA (cabotegravir; rilpivirine) extended-release injectable suspensions. oral therapy for patients who will miss planned injection dosing with CABENUVA. |
| Dosage and Administration | One tablet of VOCABRIA 30 mg taken orally once daily for approximately 1 month in combination with one tablet of EDURANT (rilpivirine) 25 mg taken orally once daily with a meal. |
| Mechanism of action | Cabotegravir is an HIV-1 antiretroviral drug.Cabotegravir inhibits HIV integrase by binding to the integrase active site and blocking the strand transfer step of retroviral deoxyribonucleic acid (DNA) integration which is essential for the HIV replication cycle. The mean 50% inhibitory concentration (IC50) value of cabotegravir in a strand transfer assay using purified recombinant HIV-1 integrase was 3.0 nM. |
| Absorption |
Tmax (h), median : 3 Effect of high-fat meal (relative to fasting): 1.14 AUC(0-inf) ratioa : (1.02, 1.28) |
| Food Effect | - |
| Distribution |
% Bound to human plasma proteins : >99.8 Blood-to-plasma ratio : 0.52 CSF-to-plasma concentration ratio (median [range])b : 0.003 (0.002 to 0.004) t1/2 (h), mean : 41 |
| Metabolism | Metabolic pathways : UGT1A1 UGT1A9 (minor) |
| Elimination |
Excretion : Major route of elimination : Metabolism % of dose excreted as total 14C (unchanged drug) in urinec : 27 (0) % of dose excreted as total 14C (unchanged drug) in fecesc : 59 (47) |
| Peak plasma time (Tmax) | 3 hours |
| Half life | 41 hours |
| Bioavailability | - |
| Age, gender | No clinically significant differences in the pharmacokinetics of cabotegravir were observed based on age, sex, race/ethnicity, body mass index, or UGT1A1 polymorphisms. The effect of hepatitis B and C virus co infection on the pharmacokinetics of cabotegravir is unknown. The pharmacokinetics of cabotegravir has not been studied in pediatric patients and data are limited in subjects aged 65 years or older. |
| DMF | Status | Type | Submit Date | Holder |
|---|---|---|---|---|
| Not Available | ||||
| Parameters | Details |
|---|---|
| Strength | 30 MG |
| Excipients used | Hypromellose, lactose monohydrate, magnesium stearate, microcrystalline cellulose, and sodium starch glycolate |
| Composition of coating material | Hypromellose, polyethylene glycol, and titanium dioxide |
| Composition of caspule shell | NA |
| Pharmaceutical Development |
The finished product is an immediate release film coated tablet for oral administration, containing 31.62 mg of cabotegravir sodium (micronised), equivalent to 30 mg of cabotegravir free acid. Vocabria tablets are white film-coated, oval-shaped tablets, (approximately 8.0 mm by 14.3 mm), debossed with “SV CTV” on one side. The finished product (FP) is packaged into opaque, white high-density polyethylene (HDPE) bottles with a polypropylene child-resistant closure that include a polyethylene faced induction heat seal liner. A science and risk-based approach, applying Quality by Design (QbD) and quality risk management (QRM) in accordance with ICH Q8, Q9, Q10, has been used to develop Vocabria film-coated tablets. The Quality Target Product Profile (QTPP) has established the desired quality characteristics of the finished product. The finished product Critical Quality Attributes (CQAs) have been identified and an understanding of the impact of the attributes of the active substance, excipients, container closure system and in-process materials, as well as the process parameters of the manufacturing process on finished product quality has been established. The knowledge gained from the pharmaceutical development and manufacturing experience have provided the scientific understanding to support the control strategy to assure product quality, which incorporates target values/set points, PARs, and a design space for the granulation unit operation. The active substance for the cabotegravir tablets is the sodium salt of cabotegravir, solid state Form 4, and is micronised to meet the QTPP of finished product. Cabotegravir sodium is classified a BCS class 2 compound (see pK report). The sodium salt has higher solubility than the free acid form ensuring oral bioavailability and appropriate pharmacokinetics. The solid-state form 4 is the most thermodynamically stable. Particle size distribution (PSD) was studied in a human pharmacokinetics study that showed that micronised cabotegravir sodium gave an increased AUC and Cmax when compared to nonmicronised substance. The PSD of cabotegravir sodium has been confirmed as an active substance CQA and ensures that dissolution profile is met. It is well controlled by the micronisation process for the active substance and is tested in the AS specification. Comparative dissolution profiles are obtained regardless of micronisation site. All chosen excipients are widely used in solid oral products and the levels chosen for this product are within typical ranges used for tablets in view of the functions stated in the composition table. There are no novel excipients used in the finished product formulation. The list of excipients is included in section 6.1 of the SmPC and in paragraph 2.1.1 of this report. The choice of AS crystal form, particle size limits, the excipients, their functions and quantity were sufficiently justified and explained. The development of the formulae used for clinical studies was described. Design of experiments (DoE) studies were performed in order to select the formulation responding to the QTPP profile. The DoE outcome determined the levels of the binder, disintegrant and drug load. The formula used in phase 2b studies had low solubility; a bio-equivalent formula has been developed for phase 3 studies and was used in all phase 3 studies, including pivotal studies, in stability studies and is the commercial formula. The development of the dissolution method has been well described. A DoE study was performed for the selection of surfactant concentration and rotation speed. The discriminatory power of the method has been sufficiently demonstrated. The manufacturing process used for Vocabria film coated tablets consists of dry mixing of intragranular excipients with the active substance, high shear wet granulation, wet screening, and fluid bed dryingfollowed by dry milling. The film-coated tablets are packaged in HDPE bottles. The selected process ensured that the finished product CQAs are met consistently. The finished product CQAs and the input materials attributes or process parameters that determine the CQA are identified. A risk assessment has been conducted and the relationship between process parameters and intermediate CQA and the finished product CQA was established. Process development has been conducted at commercial scale. The granulation unit operation has been identified to impact the CQAs. Based on the DoE, a design space was established for this step. The classification of process parameters as critical and non-critical has been supported and the proposed post authorisation management is acceptable. Vocabria film coated tablets are packed into opaque, white HDPE bottles with polypropylene child resistant closures, with a polyethylene faced induction heat seal liner. Stability studies confirmed that a desiccant in the packaging is not necessary. The HDPE is pigmented white with titanium dioxide. The plastic packaging materials components comply with the EU Commission Regulation No. 10/2011 on plastic materials and articles intended to come into contact with food. The HDPE bottles comply with Ph. Eur. 3.1.3 Polyolefines. Acceptable specifications are provided for the bottles and closures as well as Certificates of analysis from the suppliers. |
| Manufacture of the product |
The manufacturer of Vocabria film coated tablets has been stated. The manufacturing process consists of pre-mix of intra-granular components, wet granulation, screening and drying of granulate, milling, blending with extra-granular excipients, lubrication, compression, film-coating, and packaging. The manufacturing processes is a standard process. A DS is applied to the wet granulation unit operation which has been adequately discussed during pharmaceutical development studies. The critical process parameters have been identified and justified during development have been identified and justified during development. |
| Tablet / Capsule Image |

|
| Appearance | White, oval, film-coated, biconvex tablet debossed with “SV CTV” on one side |
| Imprint code / Engraving / Debossment | Debossed with “SV CTV” on one side |
| Score | No score |
| Color | WHITE |
| Shape | OVAL |
| Dimension | 14mm |
| Mfg by |
GlaxoSmithKline Research Triangle Park, NC 27709 |
| Mfg for |
ViiV Healthcare Research Triangle Park, NC 27709 |
| Marketed by | - |
| Distributed by | - |
| Application No. | Prod No | Patent No | Patent Expiration | Drug Substance Claim | Drug Product Claim | Patent Use Code | Delist Requested | Link |
|---|---|---|---|---|---|---|---|---|
| N212887 | 1 | 8410103 | January 28, 2026 | DS | DP | U-3061 | - | Download |
| N212887 | 1 | 10927129 | January 28, 2026 | DS | DP | - | - | Download |
| USP Apparatus | Speed (RPMs) | Medium | Volume (mL) | Recommended Sampling Times (minutes) | Date Updated |
|---|---|---|---|---|---|
| USP Apparatus II(Paddle) | 60±2 RPM | 0.01M HCl with 0.2% CTAB | 1000±10 mL | Q= % at 25 min | As per SBOA |
| Label | Link |
|---|---|
| FDA label | Download |
| FDA chemistry review | Download |
| FDA Pharmacology Review(s) | Download |
| FDA Clinical Pharmacology Biopharmaceutics Review(s) | Download |
| FDA BE Recommendation | |
| European Public Assessment Report | Download |
| Territory | Brand name / Generic company name | Link |
|---|---|---|
| EU | VOCABRIA | Download |
| UK | - | |
| US | VOCABRIA | Download |
| - |
| www.accessdata.fda.gov, www.drugbank.ca, www.ema.europa.eu, www.medicines.org.uk, dailymed.nlm.nih.gov |
