| Active Ingredient | AFATINIB DIMALEATE |
|---|
| Drug Name | FDA Application No. | Company | Dosage Form;Route | Strength | RLD Strength | Original Approval or Tentative Approval Date |
Exclusivity Expiration (NCE) |
Exclusivity Expiration (ODE) |
Chemical Type |
Review Classification |
Marketing Status |
TE Code |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| GILOTRIF | (NDA) 201292 | BOEHRINGER INGELHEIM | TABLET;ORAL | EQ 20MG BASE, EQ 30MG BASE, EQ 40MG BASE | EQ 40MG BASE | July 12, 2013 | July 12, 2018 | July 12, 2020 | 1 New molecular entity (NME) | P Priority review drug O Orphan drug | Prescription | None |
| Parameters | Details |
|---|---|
| Structural Formula |
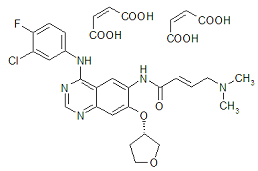 |
| Chemical Name | 2-butenamide, N-[4-[(3-chloro-4-fluorophenyl)amino]7-[[(3S)-tetrahydro-3-furanyl]oxy]-6-quinazolinyl]-4-(dimethylamino)-,(2E)-, (2Z)-2-butenedioate (1:2) |
| CAS No | 850140-72-6 |
| Molecular Formula | C32H33ClFN5O11 |
| Molecular Weight | 718.1 g/mol |
| Appearance | It exists as a salt (dimaleate) and is highly soluble in water. The highest solubility in organic solvents is observed in DMSO then in methanol. For most of the other organic solvents solubility is less than 1 mg/ml. It is highly soluble in aqueous buffer media with a pH of less than 6. The solubility of afatinib dimaleate is pH-dependent. It was noted that the free base has two ionizable groups due to presence of a dimethylamine and a quinazoline moiety. |
| Solubility | >50 mg/mL |
| Water Solubility | water soluble |
| Polymorphism | Regarding polymorphism, Forms A and B are the only under ambient conditions stable forms. Anhydrous form A and form B, do not directly transform into each other, both forms represent two totally independent crystalline modifications. Form A and form B are hygroscopic. Due to its more favourable physicochemical properties form A has been selected for development. Very similar and high solubility of forms A and B was observed under physiological conditions. |
| pKa (Strongest Acidic) | 12.49 (Predicted) |
| pKa (Strongest Basic) | 8.81 (Predicted) |
| Log P | 3.77 (Predicted) |
| Identification | IR, UV, and HPLC |
| Degradation | Degradation was seen under weak acidic to neutral media and oxidation conditions. Photostability study showed that irradiation of the active substance in the solid state stimulates isomerisation reactions. |
| Hygroscopic | hygroscopic |
| Photostability study | - |
| Melting Point | - |
| BCS Class | I or III |
| Manufacture of API | The manufacturing process consists of six main synthetic steps including formation of the dimaleate salt and its crystallisation using well defined starting materials with acceptable specifications. The S configuration of afatinib is derived from the stereochemistry of one of the starting materials. The other proposed starting materials contain no chiral centres or olefinic double bonds that could result in stereoisomers. The correct absolute configuration S of the chiral centre for afatinib has been confirmed by the analysis of enantiomeric purity (polarimetry and chiral gas chromatography). Based on the synthetic steps, it has been justified and confirmed that a racemisation or inversion cannot occur. The active substance is packaged in a closed plastic (low density polyethylene) bag, placed in a moisture-protecting aluminium laminated pouch. This pouch is heat-sealed and finally packaged into a labelled fibre drum prior to storage and shipping. |
| Parameters | Details |
|---|---|
| Indications and Usage | GILOTRIF is indicated for the first-line treatment of patients with metastatic non-small cell lung cancer (NSCLC) whose tumors have epidermal growth factor receptor (EGFR) exon 19 deletions or exon 21 (L858R) substitution mutations as detected by an FDA-approved test. Limitation of Use: Safety and efficacy of GILOTRIF havenot been established in patients whose tumors have other EGFR mutations. |
| Dosage and Administration |
Patient Selection: Select patients for the first-line treatment of metastatic NSCLC with GILOTRIF based on the presence of EGFR exon 19 deletions or exon 21 (L858R) substitution mutations in tumor specimens Recommended Dose : The recommended dose of GILOTRIF is 40 mg orally once daily until disease progression or no longer tolerated by the patient. Take GILOTRIF at least 1 hour before or 2 hours after a meal. Do not take a missed dose within 12 hours of the next dose. |
| Mechanism of action | Afatinib covalently binds to the kinase domains of EGFR (ErbB1), HER2 (ErbB2), and HER4 (ErbB4) and irreversibly inhibits tyrosine kinase autophosphorylation, resulting in downregulation of ErbB signaling. Afatinib demonstrated inhibition of autophosphorylation and in vitroproliferation of cell lines expressing wildtype EGFR or those expressing selected EGFR exon 19 deletion mutations or exon 21 L858R mutations, including some with a secondary T790M mutation, at afatinib concentrationsachieved, at least transiently, in patients. In addition, afatinib inhibited in vitroproliferation of cell lines overexpressing HER2. Treatment with afatinib resulted in inhibition of tumor growth in nude mice implanted with tumors either overexpressing wild type EGFR or HER2 or inan EGFR L858R/T790M double mutant model. |
| Absorption | Following oral administration of GILOTRIF tablets, time to peak afatinib plasma concentrations (Tmax) is 2 to 5 hours. Maximum concentration (Cmax) and area under the concentration-timecurve from time zero to infinity (AUC0-∞) values increased slightly more than dose proportional in the range of 20 to 50 mg. The geometric mean relative bioavailability of 20 mg GILOTRIF tablets was 92%as compared to an oral solution. |
| Food Effect | A high-fat meal decreased Cmaxby 50% and AUC0-∞by 39% relative to the fasted condition. |
| Distribution | In vitro binding of afatinib to human plasmaproteins is approximately 95%. Steady-state plasma concentrations are achieved within 8 days of repeatdosing of GILOTRIF resulting in an accumulation of 2.8-fold for AUC and 2.1-fold for Cmax. |
| Metabolism | Covalent adducts to proteins are the major circulating metabolites of afatinib and enzymatic metabolism of afatinib is minimal. |
| Elimination | In humans, excretion of afatinib is primarily viathe feces (85%) with 4% recovered in the urine following a single oral dose of [14C]-labeled afatinib solution. The parentcompound accounted for 88% of the recovered dose. The elimination half-life ofafatinib is 37 hours after repeat dosing in cancer patients. |
| Peak plasma time (Tmax) | 2 to 5 hours |
| Half life | 37 hours |
| Bioavailability | The geometric mean relative bioavailability of 20 mg GILOTRIF tablets was 92%as compared to an oral solution. |
| Age, gender | Based on the population pharmacokinetic analysis, weight, gender, age, and race do not have a clinically important effect on exposure of afatinib. |
| DMF | Status | Type | Submit Date | Holder |
|---|---|---|---|---|
| Not Available | ||||
| Parameters | Details | |||
|---|---|---|---|---|
| Strength | 20MG | 30MG | 40MG | |
| Excipients used | lactose monohydrate, microcrystalline cellulose, crospovidone, colloidal silicon dioxide, magnesium stearate. | |||
| Composition of coating material | hypromellose, polyethylene glycol, titanium dioxide, talc, polysorbate 80 | hypromellose, polyethylene glycol, titanium dioxide, talc, polysorbate 80, FD&C Blue No. 2 | ||
| Composition of caspule shell | - | |||
| Pharmaceutical Development |
The development included optimisation of the main process steps including dry granulation of the active substance, dry blending of the active substance (dry granulate) with the excipients, subsequent compression into tablets and finally film-coating of the tablet cores, including final drying. The tablet cores of the final formulation for all strengths are manufactured from a common blend. Thus, the different dose strengths are proportionally similar to each other. The development of the dissolution method has been discussed and the method well described. The dissolution profiles of different dosage strengths have been tested at different pH values. The solubility of afatinib dimaleate is high throughout the physiological pH range from pH 1 to 7.5. Media in this range provided sufficient solubility of the drug substance for sink conditions for the intended commercial dosage strengths 20 mg, 30 mg, 40 mg and 50 mg. The results demonstrate comparable dissolution profiles independent of the dosage strength. All four dosage strengths of the final formulation are deemed equivalent. The discriminatory power of the dissolution method has been demonstrated. The primary packaging is a blister/pouch packaging. The blister card consists of a polymer bottom foil and a lidding foil. The bottom foil is a laminate double layer foil, with an outer polyvinyl chloride (PVC) film and an inner polyvinylidene chloride(PVDC) film whereas the lidding foil is an aluminium foil. An additional pouch is used as a secondary packing system containing a molecular sieve sachet as desiccant. This pouch consists of a three-ply laminated aluminium foil, with an outer polyester (PE) film, an aluminium foil, and an inner polyethylene (PET) film. |
|||
| Manufacture of the product | The manufacturing process consists of five main steps: dry granulation, blending, tableting, filmcoating and packaging. At the end of the process, the tablets are dried to reduce residual moisture so that long-term stability of the drug product is improved. The process is considered to be a standard manufacturing process. | |||
| Tablet / Capsule Image |

|

|
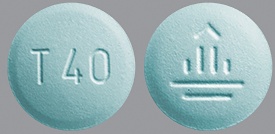
|
|
| Appearance | white to slightly yellowish, film-coated, round, biconvex, bevel-edged tablets debossed with “T20” on one side and the Boehringer Ingelheim company symbol on the other side | dark blue, film-coated, round, biconvex, bevel-edged tablets debossed with “T30” on one side and the Boehringer Ingelheim company symbol on the other side | light blue, film-coated, round, biconvex, bevel-edged tablets debossed with “T40” on one side and the Boehringer Ingelheim company symbol on the other side | |
| Imprint code / Engraving / Debossment | debossed with “T20” on one side and the Boehringer Ingelheim company symbol on the other side | debossed with “T30” on one side and the Boehringer Ingelheim company symbol on the other side | debossed with “T40” on one side and the Boehringer Ingelheim company symbol on the other side | |
| Score | no score | no score | no score | |
| Color | white to slightly yellowish | dark blue | light blue | |
| Shape | round, biconvex, bevel-edged | round, biconvex, bevel-edged | round, biconvex, bevel-edged | |
| Dimension | 8mm | 9mm | 10mm | |
| Mfg by | Boehringer Ingelheim Pharmaceuticals, Inc. (US, EU) | |||
| Mfg for | - | |||
| Marketed by | Boehringer Ingelheim Pharmaceuticals, Inc. (US, EU) | |||
| Distributed by | Boehringer Ingelheim Pharmaceuticals, Inc. (US, EU) | |||
| Application No. | Prod No | Patent No | Patent Expiration | Drug Substance Claim | Drug Product Claim | Patent Use Code | Delist Requested | Link |
|---|---|---|---|---|---|---|---|---|
| N201292 | 1 | 6251912 | July 29, 2018 | Y | Y | U - 1067 | - | Download |
| N201292 | 1 | 8426586 | October 10, 2029 | Y | Y | - | - | Download |
| N201292 | 1 | 8545884 | December 19, 2029 | - | Y | - | - | Download |
| N201292 | 1 | RE43431 | January 22, 2022 | Y | Y | - | - | Download |
| USP Apparatus | Speed (RPMs) | Medium | Volume (mL) | Recommended Sampling Times (minutes) | Date Updated |
|---|---|---|---|---|---|
| II (Paddle) | 75 | McIlvaine Buffer pH 4.0 | 900 | 5, 10, 15, 20 and 30 | May 28, 2015 |
| Label | Link |
|---|---|
| FDA label | Download |
| FDA chemistry review | Download |
| FDA Pharmacology Review(s) | Download |
| FDA Clinical Pharmacology Biopharmaceutics Review(s) | Download |
| FDA BE Recommendation | Download |
| European Public Assessment Report | Download |
| Territory | Brand name / Generic company name | Link |
|---|---|---|
| EU | GIOTRIF | Download |
| UK | GIOTRIF | Download |
| US | GILOTRIF | Download |
| 50MG strength of Afatinib dimaleate tablet (GIOTRIF) is also approved in EU and UK. |
| www.accessdata.fda.gov, www.drugbank.ca, www.ema.europa.eu, www.medicines.org.uk, dailymed.nlm.nih.gov |
